A good example of this effect would be silver, as its light isotopes use electron capture and the heavier ones decay by negative beta emission. Anomalies in elemental distributions are thought to be partly a result of this effect on electron capture.Ĭhemical bonds can also affect the rate of electron capture to a small degree (generally less than 1%) depending on the proximity of electrons to the nucleus.Īround the elements in the middle of the periodic table, isotopes that are lighter than stable isotopes of the same element tend to decay through electron capture, while isotopes heavier than the stable ones decay with a process called negative beta decay. Of the three modes, this invention relates about the phenomenon of electron capture but not through the decay process of an atom. 
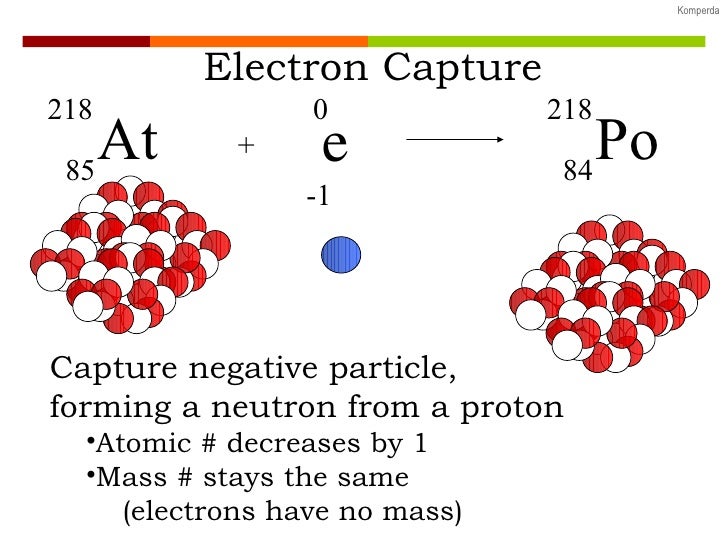
It is hypothesized that such elements, if formed by the r-process in exploding supernovae, are ejected fully ionized and so do not undergo radioactive decay as long as they do not encounter electrons in outer space. And example of this would be is Be-7 undergoes electron capture. (Please note that it is one of the initial atom's own electrons that is captured, not a new, incoming electron as might be suggested by the way the above reactions are written.) Radioactive isotopes which decay by pure electron capture can, in theory, be inhibited from radioactive decay if they are fully ionized ("stripped" is sometimes used to describe such ions). When transiting to the ground state, the atom will emit an X-ray photon (a type of electromagnetic radiation) and/or Auger electrons. The atom moves into an excited state with the inner shell missing an electron. This is exhibited in the potassium-argon decay. By changing the number of protons, electron capture transforms the nuclide into a new element. Question: Which of the following reactions is an example of K-electron capture A.18373 Ta rightarrow18376W + e B.4120Ca +0-1e rightarrow4119 K. Electron capture: A parent nucleus may capture one of its own electrons and emit a neutrino. Since the proton is changed to a neutron, the number of neutrons increases by 1, the number of protons decreases by 1, and the atomic mass number remains unchanged. In electron capture, an electron from an inner orbital is captured by the nucleus of the atom and combined with a proton to form a neutron. One example of electron capture involves beryllium-7.In this case, one of the orbital electrons, usually from the K or L electron shell ( K-electron capture, also K-capture, or L-electron capture, L-capture), is captured by a proton in the nucleus, forming a neutron and a neutrino. Often the nucleus exists in an excited state as well and emits a gamma ray as it transitions to the ground state of the new nuclide. This nuclear chemistry video tutorial provides a basic introduction into radioactive decay such as alpha decay, beta decay, gamma decay, electron capture and. (We pronounce the name as "oh zhay", not "aw ger). 
As a result, a proton transforms into a neutron, and a neutrino is. We call this ejected electron an Auger electron after one of its discoverers, Pierre Victor Auger. Electron capture happens when an unstable nucleus absorbs an inner orbital electron. As you peruse these examples, notice the mass-balance of the nuclear equations (both mass number and atomic number). All it does is carry away excess energy.Īnother electron may absorb excess energy and leave the atom. Radioactive Decay Processes There are various types of decay processes that radioactive (unstable) nuclei may undergo to increase their stability. When an atom has a huge atomic mass and is deficient in neutrons inside its nucleus, it is most likely to undergo electron capture, an example of a. Many people omit the neutrino in the equation, because it has no mass or charge. Researchers have found convincing evidence for an electron-capture supernova, a stellar explosion ignited when atomic nuclei sop up electrons within a star’s core. The excess energy leaves as an X-ray photon and a neutrino. The inner shell is missing an electron.Īn outer shell electron then drops to a lower energy level to replace the missing electron. The activity of a sample of gas obtained from a basement containing radon-222 was found to be 8 pCi/L. The atom stays neutral in charge, but it now exists in an excited state. The captured electron often comes from the K-shell because those electrons are closest to the nucleus. Since a proton becomes a neutron, the number of protons decreases by 1, but the atomic mass stays the same. The electron combines with a proton to make a neutron and emits an electron neutrino.Įlectron capture is a major decay mode for isotopes with too many protons in the nucleus. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
